
NCBiotech-Funded Microscope Sheds UNC Light on Brain Circuitry of Binge Eating

-- Megan May, UNC Research
Ever wondered why you can’t eat just one? Be it chips or chocolate – or drugs or alcohol – neuroscientists have spent decades trying to uncover the reasons why people binge.
With support from a $200,000 instrumentation grant from the North Carolina Biotechnology Center in 2016, researchers at the University of North Carolina at Chapel Hill have discovered a brain circuit about hedonic versus homeostatic feeding in mice. That is, what drives them to binge eat calorie-rich food that tastes good (hedonic) versus what drives them to eat only in response to hunger (homeostatic).
This research is part of the overarching work to shed new light on the mechanisms of overeating and to identify potential therapeutic targets in the treatment of obesity -- and potentially in areas such as substance abuse, said Thomas Kash, Ph.D.
Kash, the John R. Andrews Distinguished Professor of Pharmacology at the UNC School of Medicine, and J. Andrew Hardaway, Ph.D., research assistant professor of pharmacology, led the study. The results were published in the journal Neuron. Both scientists are also members of the UNC Bowles Center for Alcohol Studies.
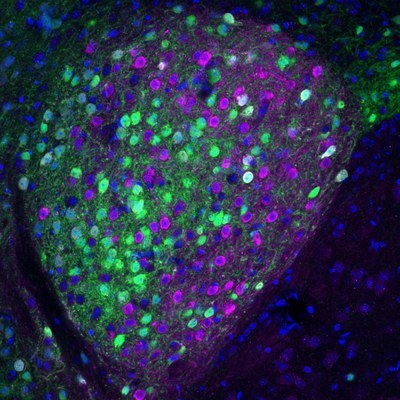
and PKC delta (magenta) neurons. -- UNC photos
And shed light it did. The researchers engineered a line of mice to make certain cells express a protein that glows red. Then they chemically “cleared” the whole mouse brains. This process removed the lipids, or fat molecules, and left the proteins, which allowed the light from the LaVision BioTec “light sheet” UltraMicroscope II to get deeper into the brains as it scanned. The result was 3D images to which they applied an algorithm that counted the cells and mapped their locations in the brains.
What they found when they followed the specialized proteins called nociceptin circuits, which previous research had led them to study further, was that the process of hedonic eating originates in the central amygdala, the part of the brain where emotion processing happens. Research has well-established that the amygdala is tied to pain, anxiety and fear (the fight-or-flight response to stressors), but this study highlights the role of it in regulating pathological eating.
When they eliminated half of the nociceptin-making neurons in the mice, the rodents’ bingeing behavior reduced and they kept their weight in check, even with access to richer, tastier food, without affecting their intake of regular food.
The UNC scientists believe this process has evolutionary roots from a primitive time of feasting and famine cycles that signaled the brain to fill up on high-calorie food whenever it was available to carry us through the periods of low- or no-availability of sustenance.


According to Kash, access to the light sheet microscope is what enabled this groundbreaking research.
“By the way the light sheet microscope works, we can scan through the entire brain in a relatively short period of time,” said Kash. “Essentially, we never would have been able to do this study without the microscope.”
Kash explained that heretofore they would make informed hypotheses about what is happening in certain regions of the brain, based upon others’ findings about various activity in certain brain regions. However, this instrument scanning the entire brain allows for scientific discovery in new ways.
“What the light sheet microscope has done is given us the ability to step back and say, ‘What is happening at this point in time across the entire brain? What looks interesting?’ Generate new hypotheses and work from there. So, we’re pretty jazzed,” said Kash.
However, he said this study didn’t even use some of the most advanced features of this model of light sheet microscope.
A "game changer" in North Carolina
“This microscope is unique in its capabilities and it’s transformative in what research it can support,” said Pablo Ariel, Ph.D., director of the microscopy services laboratory at UNC’s Department of Pathology and Laboratory Medicine, who has published a user’s guide for the microscope.
“The reason this is transformative is the way you used to do these big organ surveys you would have to physically cut the brain, stain every little slice of the brain, and then reconstruct that in 3D. That is very expensive. It is very slow. And, it is very difficult. So, now the whole process is much faster, it’s much cheaper and it’s much easier. Doing that before this microscope was basically impossible.” he said. In addition, he said it can accommodate samples that have been cleared (made transparent) with techniques that use harsh solvents, which you cannot use with other microscopes.
The model of this instrument at UNC is also the only one of its kind in North Carolina, said Ariel. In addition to serving UNC investigators, researchers travel from Duke University and North Carolina State University and other institutions to use it, as well as from several biomedical companies. Ariel said he welcomes commercial use of the microscope and wants to see it utilized fully by anyone whose work it will support.
There are other light sheet microscopes in the state, he said, but the UNC device is the only one that is fully compatible with organic solvents, which, he believes, are what enable the techniques that lead to the best clearing of the brain. Also, the scope in Chapel Hill can accommodate large samples. And it can also accommodate other organs, like a heart or liver, for applications in a wide range of research using mice, which he said are a very biomedically relevant species.
“Outside of this project it’s been a real game-changer for how we approach problems and understand how the brain works,” said Kash, who also has other projects using data from this microscope, in combination with mouse brain data collected from magnetic resonance imaging, to try to answer even more big neuroscience questions about the activities of microscopic particles.
