
Bryn Pharma’s Anaphylaxis Treatment Gains $11M in Latest Fundraising Round

Many investors have a great deal of confidence in Bryn Pharma, a Raleigh-based pharmaceutical company that is developing an epinephrine nasal spray to treat anaphylaxis.
In fact, they invested $11 million in the company’s latest fundraising round, according to a filing with the U.S. Securities and Exchange Commission. This was an extension of the October 2019 financing bringing the total in that series to $26 million.
The company’s first reported raise was for $9.2 million in July 2018. It raised $17.2 million in February 2019 and $15 million in October 2019.
It is estimated that 49 million Americans with severe, possibly life-threatening allergies are at risk for anaphylaxis. For 30 years, the standard of care has been for patients to carry two epinephrine auto-injectors with them at all times. Studies show, however, that less than half of patients diagnosed with anaphylaxis carry one auto-injector and a much smaller percentage carry the recommended two.
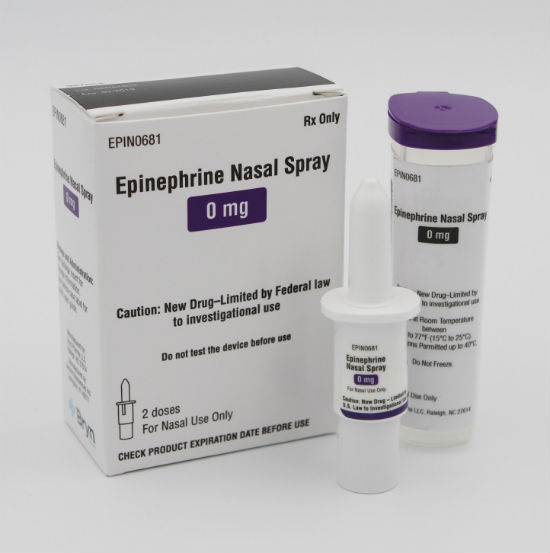
Bryn Pharma’s Bryn Intranasal Epinephrine Spray (BRYN-NDS1C) is a needle-free, portable and easy-to-use device for administering epinephrine. The U.S. Food and Drug Administration granted BRYN-NDS1C Fast Track designation early last year.
Investors aren’t the only ones recognizing BRYN-NDS1C’s promise. Over the past few months, three peer-reviewed scientific journals have published the results of preclinical studies in support of the device – Respiratory Research, Pharmacology Research and Perspectives and Pharmaceutical Research.
In April 2020, results of the latest human clinical trial were presented at the American Academy of Asthma, Allergy and Immunology annual meeting. Trial researchers concluded that intranasal delivery of epinephrine from the needle-free, bi-dose device was comparable to intramuscular injection in pharmacokinetics, pharmacodynamics and safety.
More human clinical trials continue as the company moves closer to commercialization.
