
Raleigh’s UVision360 Gets FDA OK For Biopsy Sheath

A new biopsy sheath developed by Raleigh med tech startup UVision360 is ready to hit the market after landing federal approval this week.
UVision360 announced it had received the greenlight from the U.S. Food and Drug Administration for its patented LUMINELLE 360° Bx Sheath.
The device expands the startup’s “all-in-one” gynecological system LUMINELLE DTx, an endoscopic platform that enables diagnostic and operative procedures, such as uterine and bladder biopsies, to be performed in the office. The goal is to make them more accessible and affordable.
The new sheath, the company says, makes the system the “first and only” integrated biopsy device not requiring the use of additional instruments.
"The FDA approval of our LUMINELLE Bx Sheath not only marks a major milestone for UVision360, but is also a game changer when it comes to the standard of care for tissue biopsy procedures," said UVision360 CEO Allison London Brown. "The affordability, reusability, and adaptability of the [system] enhances the patient experience and gives doctors the flexibility to perform needed procedures when and where they see fit."
"Prior to [this], doctors could not visualize and sample abnormal tissue without the use of additional biopsy tools," added Dr. David Robinson, UVision360's chief medical officer.
"Now, doctors will be able to easily visualize areas of concern, while sampling occurs. In addition, the clear biopsy sampling tube allows the physician to see how much actual tissue they are acquiring, ensuring they have an adequate sample.”
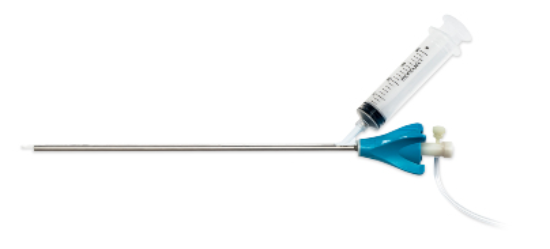
The company says it will bring the sheath to market with a limited launch in the coming months. It plans a broader distribution “shortly thereafter.”
Back in 2016, Brown, a North Carolina State University grad, founded UVision360 with the mission to advance access to care by transitioning procedures from the operating room to the physician’s office.
The startup received FDA approval for its LUMINELLE DTx System in 2018. The simplified, compact, and modular endoscopic system allows gynecologists to perform hysteroscopic procedures with “superior visualization” due to its high-definition optics and integrated flow system.
In 2016 the North Carolina Biotechnology Center awarded UVision360 a $250,000 Small Business Research Loan to help advance the important technology. Two years later, it received another $250,000 loan.
According to Crunchbase, the company has raised a total of $8.1 million to date.
