
Nearly $5 Million in Cash Will Get Cary Medical Device to European Approval

The nearly $5 million in equity that Cary-based Phoenix Cardiac raised this month should enable it to gain European approval of its medical device that fixes a heart valve problem, according to its founder and CEO, Gopal Muppirala, MBA.
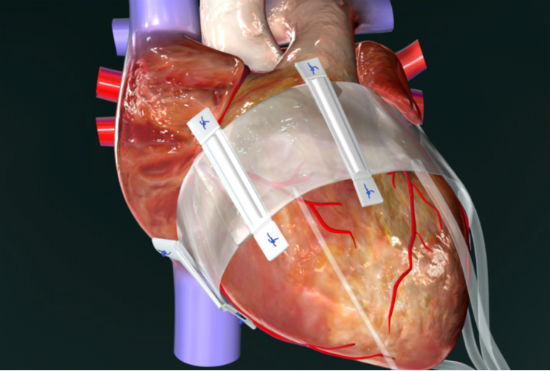
The company’s BACE device (an acronym for Basal Annuloplasty of the Cardia Externally) offers an “easier, safer, and simpler” way to treat functional mitral valve regurgitation, Muppirala said in an interview with the North Carolina Biotechnology Center.
Currently, open-heart surgery is the standard of care to correct the valve problem, in which blood flows back into the upper chamber of the heart instead of circulating.
Less blood circulation means less oxygen to the body, and that causes breathlessness, tiredness and eventually heart failure, Muppirala explains.
-- Phoenix Cardiac photos
About six million Americans a year have mitral regurgitation (MR). It has various causes and the BACE device will be able to treat about 50 percent of sufferers, resulting in a $1.5 to $2 billion market for the device, Muppirala said.
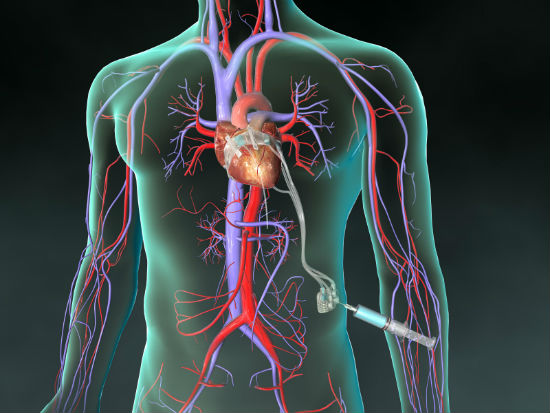
The BACE device is placed outside the heart, so open heart surgery is not necessary, eliminating its associated problems. It eliminates the need for patients to be attached to a heart bypass machine during surgery and the danger of blood clots requiring taking blood thinners for the rest of their lives.
“It offers many benefits to the patient,” Muppirala said. “Our device sits outside the heart, and makes sure blood flows in only one direction, not back into the upper chamber of the heart.” In addition, the device can be adjusted on an outpatient basis in a cariologist’s office.
Muppirala founded the virtual company with his partner, cardiac thoracic surgeon Jai Raman, in 2012. Phoenix raised $2.1 million last year. The money from both financings came from friends, family and angel investors, he said.
Being a virtual company allowed it to operate without spending a lot of cash, he added. “It’s a non-traditional business model without a lot of overhead or upkeep compared to the traditional company with an office and 15 to 20 employees.”
Phoenix Cardiac employs five and does not plan additional hiring. Raman is based in Portland, Oregon and his engineering is done in New Hampshire, but the company will remain located in the Triangle, where Muppirala has lived for 30 years.
Muppirala said development took quite a while and included trying making the device from various materials such as polyester before settling on silicone, which doesn’t stick to the heart so the device can be removed at any time. The company maintains a brief video explanation of its device on its website.
Recruiting patients for clinical trials has also been time-consuming, but the company expects to complete recruiting for a trial and applying for European approval within a few months, he said.
The company will seek U.S. Food and Drug Administration approval after achieving it in Europe. It will seek a large medical device partner to market the device rather than raising additional cash, Muppirala said. “The money we raised will take us all the way to European approval but we’re hoping a big company will take us to the next stage."
