
Baebies Wins FDA OK for Newborn Screening System
By Barry Teater, NCBiotech Writer
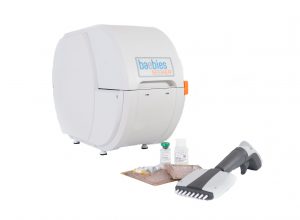 |
| The Baebies SEEKER diagnostic system. -- Baebies photo |
Durham-based Baebies Inc. has received clearance from the U.S. Food and Drug Administration to begin marketing a system to screen newborns for four rare but potentially fatal diseases.
The system, called SEEKER, measures the activity of four enzymes in dried blood spot samples that can indicate various lysosomal storage diseases (LSDs).
LSDs are inherited metabolic disorders caused by defects in the function of lysosomes, sacs of enzymes within cells that digest large molecules into fragments that the cell can use. If one of the key enzymes is defective due to a genetic mutation, the large molecules go undigested and pile up in the cell, becoming toxic.
Newborn screening, widely recognized as one of the most successful public health programs in the U.S., ensures that all babies are screened at birth for certain serious conditions. The program started more than 50 years ago and has since expanded to include more than 30 metabolic and genetic diseases.
“Although these LSDs have FDA-approved therapies, this is a significant milestone for the LSD community, as SEEKER is the first FDA-cleared product for LSD testing,” Vamsee Pamula, president and co-founder of Baebies, said in a news release. “FDA’s clearance of SEEKER resulted from a thorough and rigorous review. We look forward to providing SEEKER kits to implement safe, effective, easy and reliable screening for LSDs.”
Reduced enzyme activity indicates disease
SEEKER measures the activity of lysosomal enzymes α-L-iduronidase (IDUA), α-D-glucosidase (GAA), β-glucocerebrosidase (GBA) and α-D-galactosidase A (GLA). Reduced activity of these enzymes may indicate the diseases mucopolysaccharidosis type I (MPS-1), Pompe, Gaucher or Fabry, respectively.
These four diseases occur in about one of every 1,500 to 185,000 newborns and children, according to the U.S. Department of Health and Human Services’ Advisory Committee on Heritable Disorders in Newborns and Children. If not detected and treated, they may cause organ damage, neurological disability or death.
The recent addition of Pompe disease and MPS I to the Recommended Uniform Screening Panel (RUSP) have brought more recognition to the benefit of early detection through newborn screening for LSDs, Baebies said.
“As LSDs are being added to the RUSP, state newborn screening programs are making plans to begin screening,” said Bradford Therrell Jr., Ph.D., director of the National Newborn Screening and Global Resource Center. “FDA clearance enables state laboratories to confidently implement testing while minimizing the validation effort, resulting in a cost-effective implementation.”
LSD screening in all newborns is mandated in several states, including Arizona, Illinois, Kentucky, Michigan, Missouri, New Jersey, New Mexico, New York, Ohio, Pennsylvania and Tennessee, the FDA said in a news release. “However, until today there were no FDA-authorized devices for screening of these disorders.”
Equity funding preceded by NCBiotech support
Baebies raised $13 million in equity financing in 2015 to commercialize its newborn-screening technology. Investors include Rex Health Ventures, DUMAC, Cunning Capital, Triad LLC, the Duke Angel Network, Charleston Angel Partners and members of Baebies’ executive team.
The financing followed a $500,000 Strategic Growth Loan from the North Carolina Biotechnology Center in 2014.
Baebies licensed its microfluidics technology from Illumina, a global genetic sequencing and genotyping company based in San Diego. Illumina, which has an equity stake in Baebies, acquired the technology from Morrisville-based Advanced Liquid Logic in 2013 for $96 million.
Advanced Liquid Logic was a Duke University spinout company co-founded by Pamula and led by CEO Richard West, so the technology has come full circle with Baebies.
