
410 Medical Raises $6.3 Million in Venture Capital

Durham-based 410 Medical, a medical device company developing new technologies for emergency and critical care, has closed on a $6.3 million round of venture capital.
The Series B round was led by Hatteras Venture Partners of Durham with participation by WakeMed of Raleigh, OSF Ventures of Peoria, Ill., and Florida-based Orlando Health Ventures.
The company will use the money to develop LifeFlow, a hand-held device with a trigger for quickly infusing blood and intravenous fluids in critically ill patients.
“This new funding will be instrumental in accelerating the development of
“Over the past three years, in spite of the challenges that COVID-19 presented, we have launched LifeFlow PLUS for rapid blood and IV fluid delivery and significantly grown our user base to more than 130 hospitals and EMS agencies across the U.S.,” Chenet said.
Device used in sepsis, trauma cases
In sepsis, trauma, and other conditions that lead to hypotension and shock, rapid delivery of blood or IV fluid is a life-saving therapy, but traditional infusion methods often fail to achieve published fluid-delivery guidelines, according to 410 Medical. The LifeFlow device was designed to be a faster, simpler and more portable alternative.
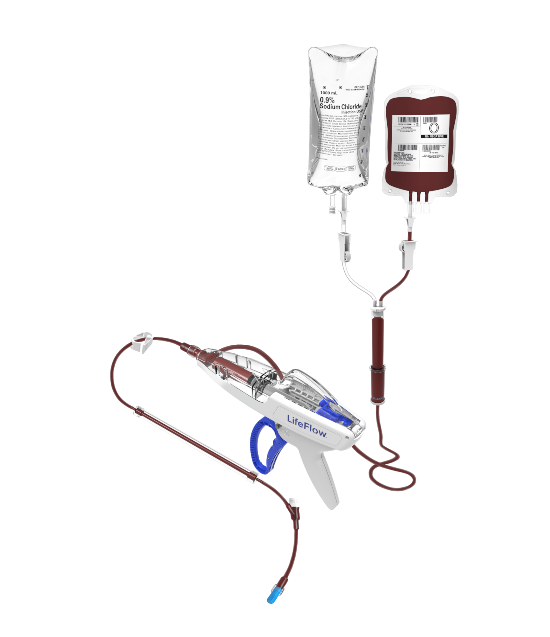
Since gaining clearance by the U.S. Food and Drug Administration in 2016, LifeFlow has been adopted at many U.S. healthcare systems, including Orlando Health.
“At Orlando Health our mission is to improve human health by supporting innovative
healthcare products and services,” said Bobby Helmadag, managing director at Orlando Health Ventures, the venture arm of Orlando Health, a not-for-profit healthcare system serving the Southeast. “With clear clinical support from our physicians, we are excited to partner with 410 Medical to implement LifeFlow at Orlando Health while supporting expanded use throughout the U.S.”
This summer, 410 Medical received up to nearly $2 million in U.S. military funding to develop two new versions of LifeFlow.
The company received an Air Force AFWERX Phase II SBIR Award of up to $750,000 to develop LifeFlow Ultra, for use when space, weight and power supply are important factors.
The company was awarded a U.S. Army Medical Research and Development Command contract of up to $1.2 million to develop LifeFlow ARS, a ruggedized version for use in austere environments and include advanced features, including automation, designed to improved combat casualty care.
Final funding amounts for both awards will be tied to technical progress.
Early funding from NC Biotech
410 Medical was founded by Raleigh physician Mark Piehl and venture capitalist Luke Roush in 2013 to develop innovative products to help emergency clinicians provide better care for critically ill patients. Piehl, who is 410’s chief medical officer, is a pediatric intensivist at WakeMed, where he was medical director of WakeMed Children’s Hospital from 2009 to 2015.
In 2016 the company was awarded a $250,000 Small Business Research Loan by the North Carolina Biotechnology Center. The loan program supports business inception and research critical to developing products, processes or tools with clear commercial potential.
410 Medical raised $3.1 million in financing in 2018 to commercialize LifeFlow. North Carolina investors in the funding round included WakeMed Health & Hospitals and the North Carolina Venture Capital Multiplier Fund, managed by Hatteras Venture Partners.
The company lists 17 employees on its website.
410 Medical’s name is derived from the calculation of a fluid-delivery goal in treating pediatric septic shock.
