
United Therapeutics Gets FDA OK for Lung Drug Delivery System

United Therapeutics Corp. has received approval from the U.S. Food and Drug Administration to begin marketing and selling an implantable system for delivering Remodulin, its treatment for pulmonary arterial hypertension (PAH), or high blood pressure in the lungs.
The system will be commercialized by a team at the company’s Research Triangle Park site, a multi-purpose facility that includes manufacturing, research and administrative functions and employs 363 people, a company spokesman said.
Remodulin is a formulation of treprostinil, a prostacyclin drug that opens constricted blood vessels. The FDA approved Remodulin two decades ago to treat PAH using external infusion pumps and catheters.
The new delivery system, developed in collaboration with Medtronic, a global medical technology company based in Dublin, avoids the use of external pumps and catheters.
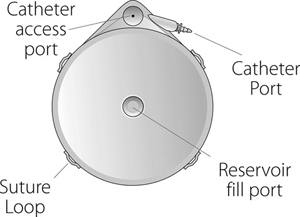
-- Medtronic illustration
The system is implanted into the body and can be refilled with Remodulin by healthcare professionals using a syringe needle through the patient’s skin. Patients can go as long as four months between refills, based on their doses of the drug.
"External infusion pumps have been used to deliver prostacyclins for PAH, but managing the therapy places a significant burden on patients, interferes with their daily activities, and runs a high risk of infections," said David Steinhaus, M.D., general manager of Medtronic’s heart failure business, part of the company’s cardiac and vascular group. "This fully implantable drug delivery system was designed to address these serious patient-care concerns."
Remodulin is one of four FDA-approved PAH drugs sold by United Therapeutics. The others are Tyvaso, Orenitram and Adcirca. The company also markets Unituxin, a drug for treating neuroblastoma, a cancer of the adrenal glands.
United, headquartered in Silver Spring, Md., was founded in 1996 and became a publicly traded company in 1999. Its shares are traded on the Nasdaq stock exchange under the ticker symbol “UTHR.”
